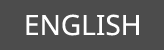天然物の全合成
当研究室では天然有機化合物の全合成に挑戦しています。合成目標とする化合物の中には、天然から僅かしか得られない希少なもの、有用かつ強力な生理活性を持つもの、あるいは珍しい構造をもつもの、ときには非常に不安定で取り扱いにくいものなどもあります。我々は、既存の方法論の組み合せでは合成困難、あるいは非常に手間がかかる化合物を積極的に取り挙げ、その全合成に挑戦しています。それらをどのような作戦で効率的かつ選択的に作り上げるか。そこが我々の腕の見せ所ともいえます。以下には、これまで私たちが行ってきた天然物合成の一例を示しました。どれも自身作です。是非ご覧下さい。
- mycinamicin IV
- vineomycinone B2
- aquayamycin
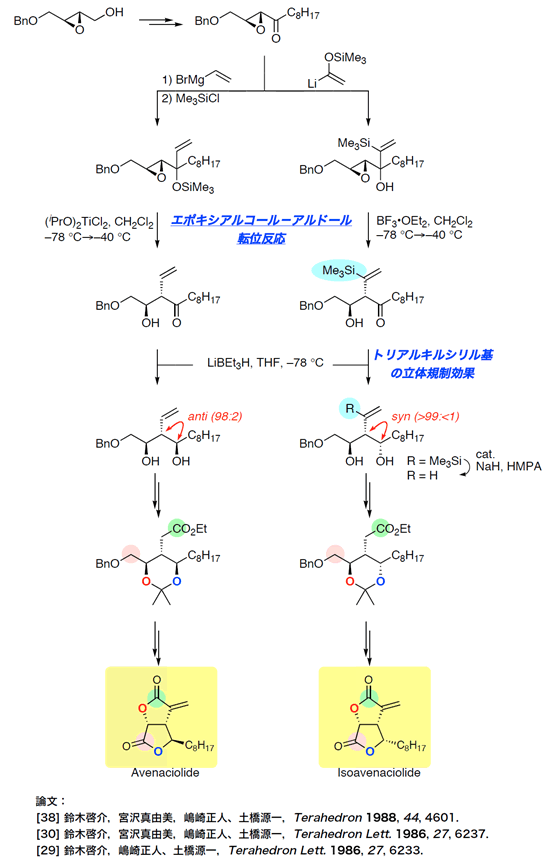
- avenaciolide
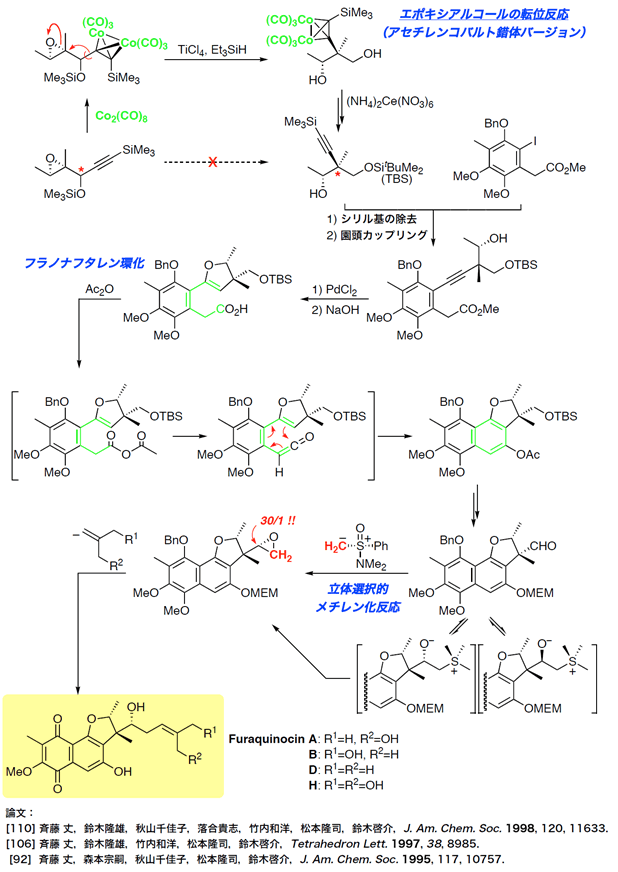
- fraquinocins
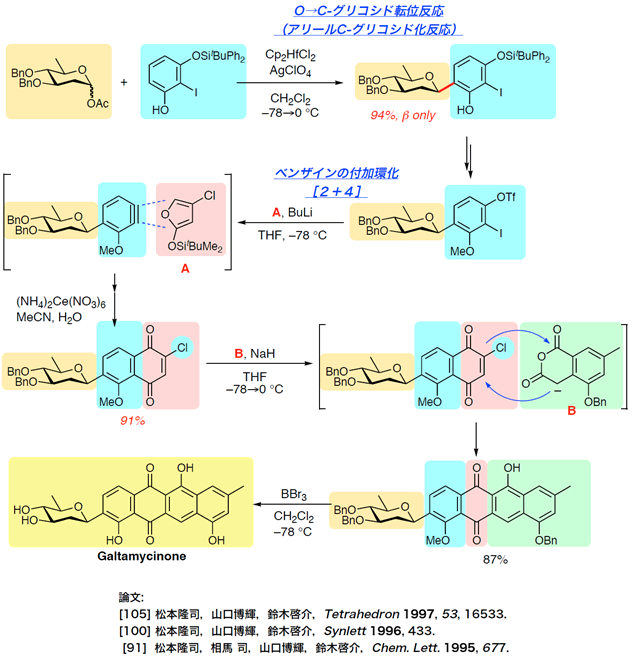
- galtamycinone
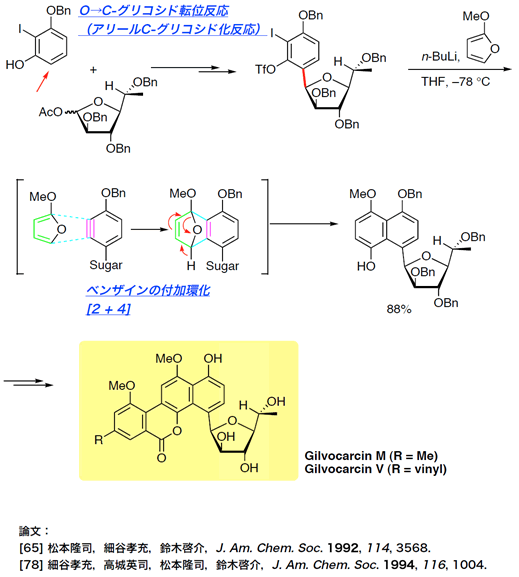
- gilvocarcins
- C104 antibiotic
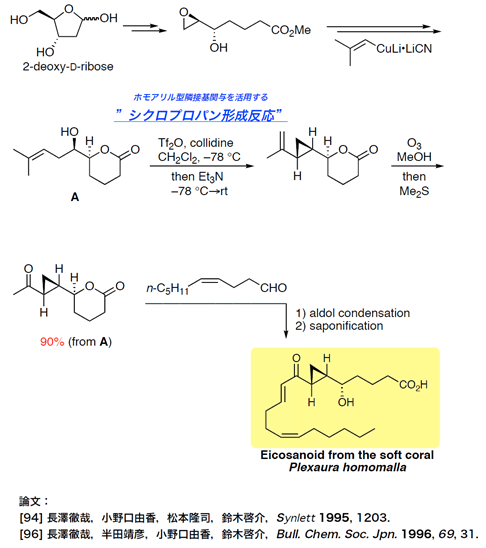
- eicosanoid
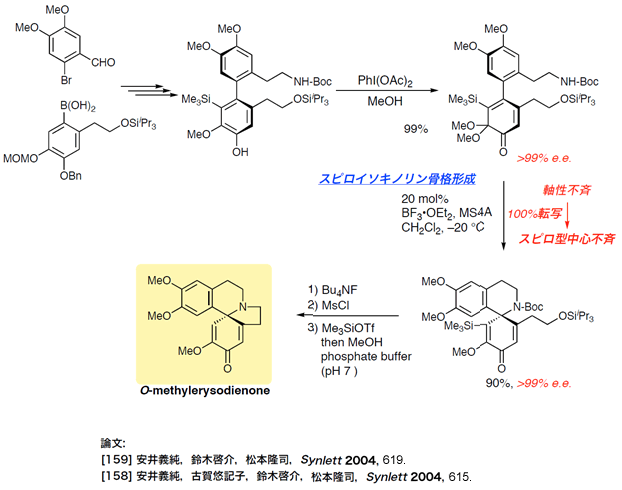
- methylerysodienone
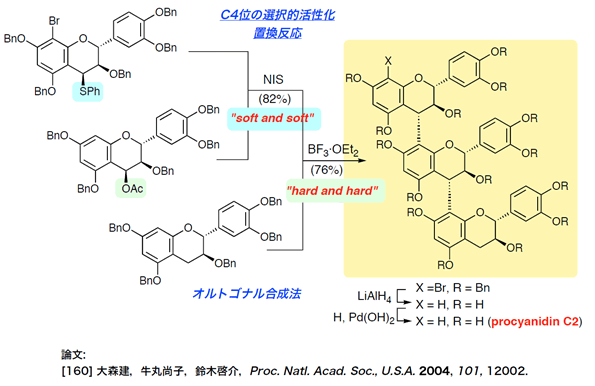
- procyanidins
- lotthanongine
- malyngolide
- seragakinone A
- BE-43472B
- saptomycin B
- cavicularin
- cinnamtannin B1
Total Synthesis of Mycinamicin IV
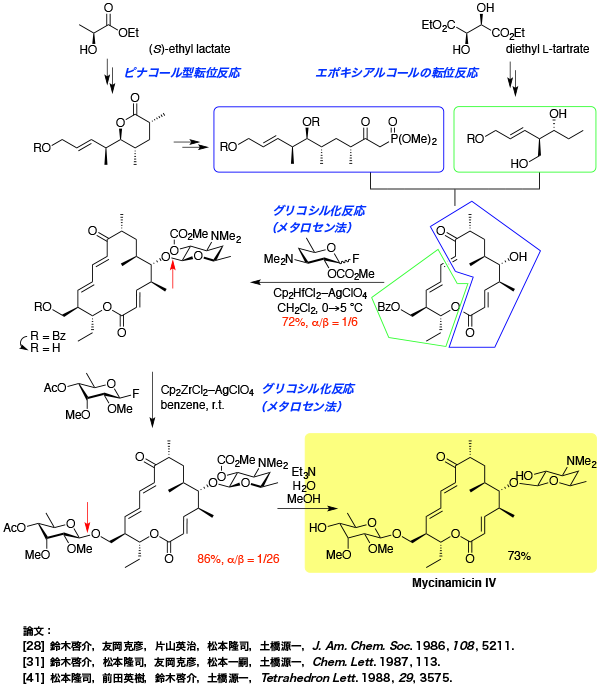
Total Synthesis of Vineomycinone B2
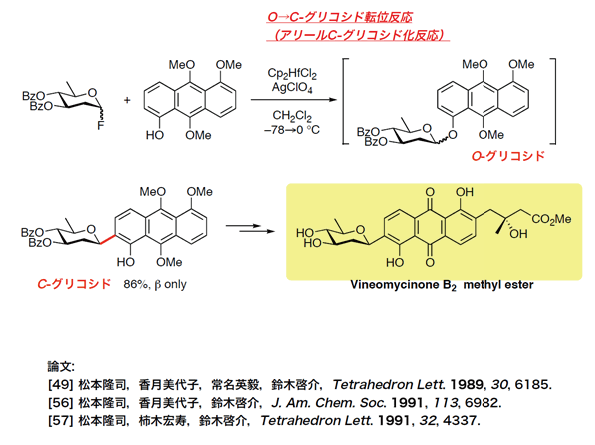
Total Synthesis of Aquayamycin
Total Synthesis of Avenaciolide
Total Syntheses of Fraquinocins
Total Synthesis of Galtamycinone
Total Syntheses of Gilvocarcins
Total Synthesis of C104 antibiotic
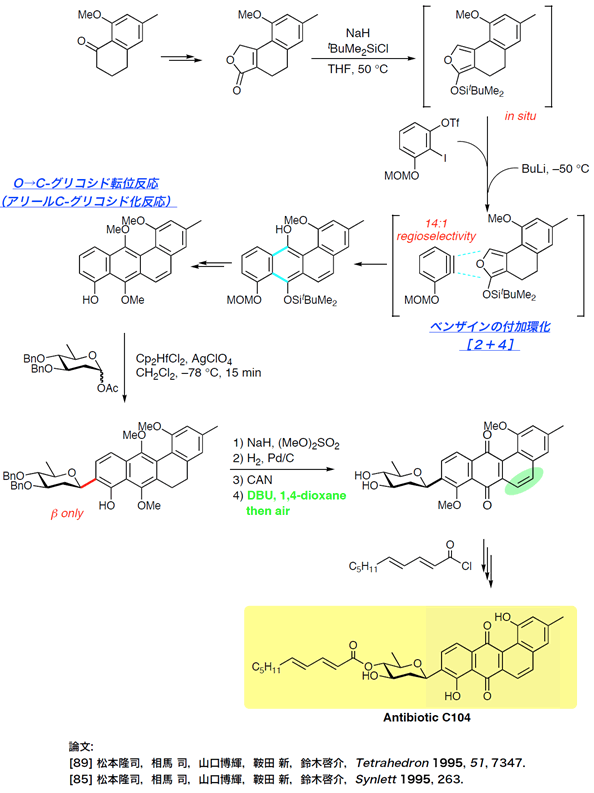
Total Synthesis of a Marine Eicosanoid
Total Synthesis of Methylerysodienone
Total Synthesis of Procyanidin
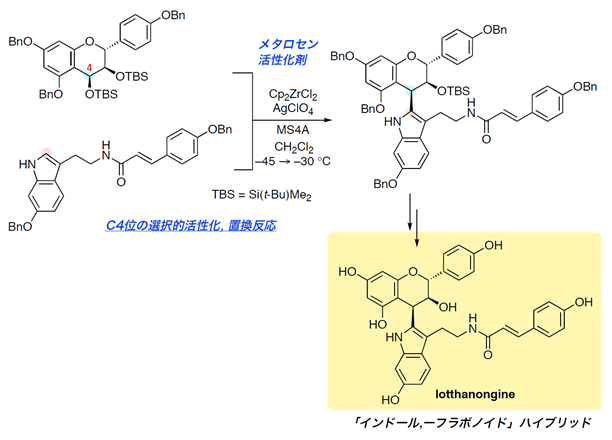
Total Synthesis of Lotthanongine
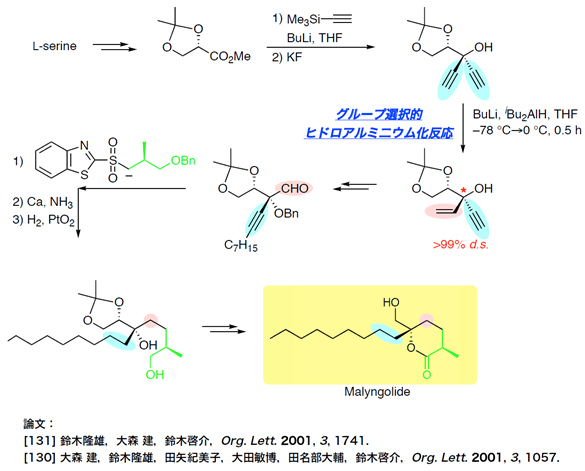
Total Synthesis of Malyngolide
Total Synthesis of Seragakinone A
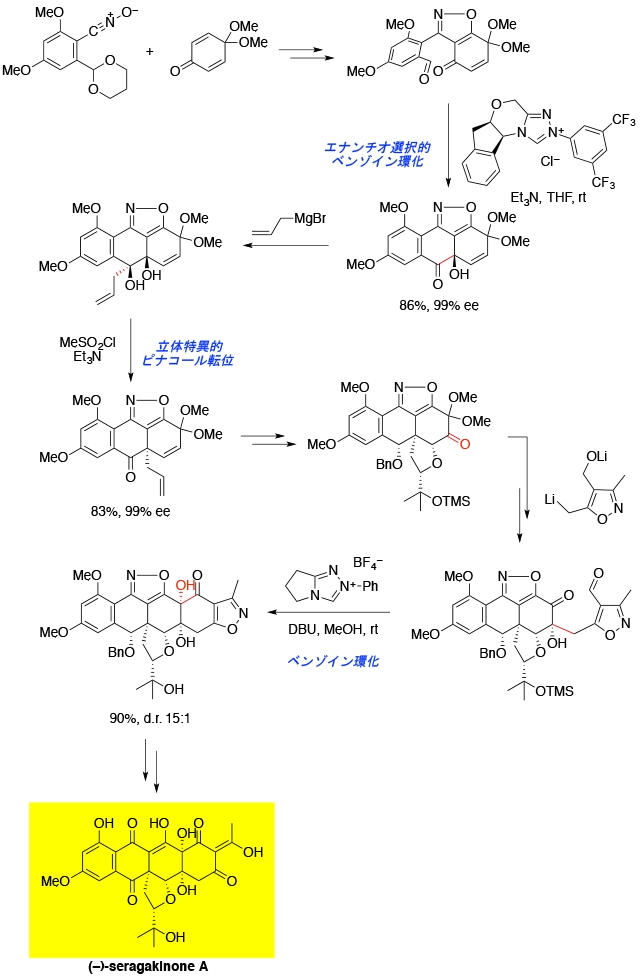
[212] A. Takada, Y. Hashimoto, K. Hikita, H. Takikawa, and K. Suzuki. Angew. Chem. Int. Ed. 2011, 50, 2297.
Total Synthesis of BE-43472B
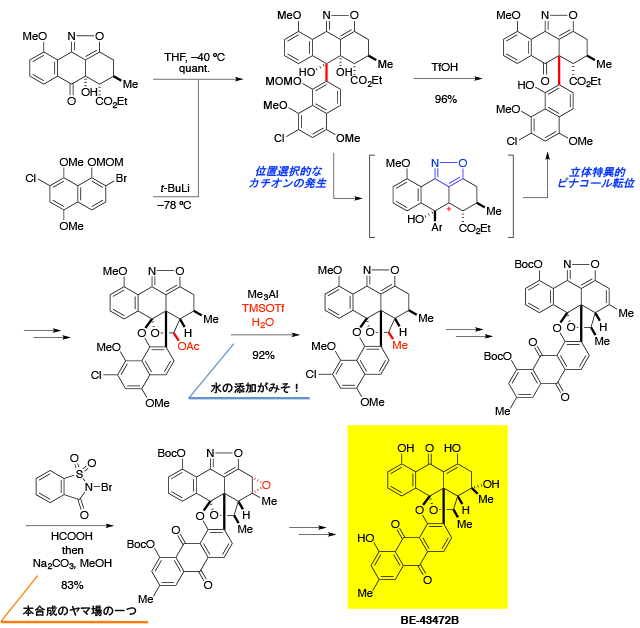
[232] Y. Yamashita, Y. Hirano, A. Takada, H. Takikawa, K. Suzuki. Angew. Chem. Int. Ed. 2013, 52, 6658.
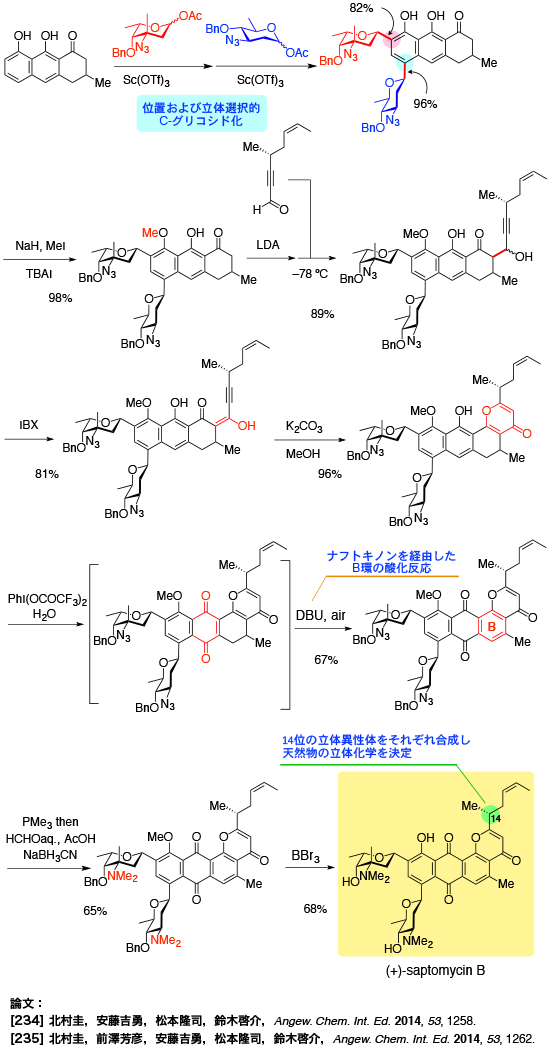
Total Synthesis of Saptomycin B
Total Synthesis of Cavicularin
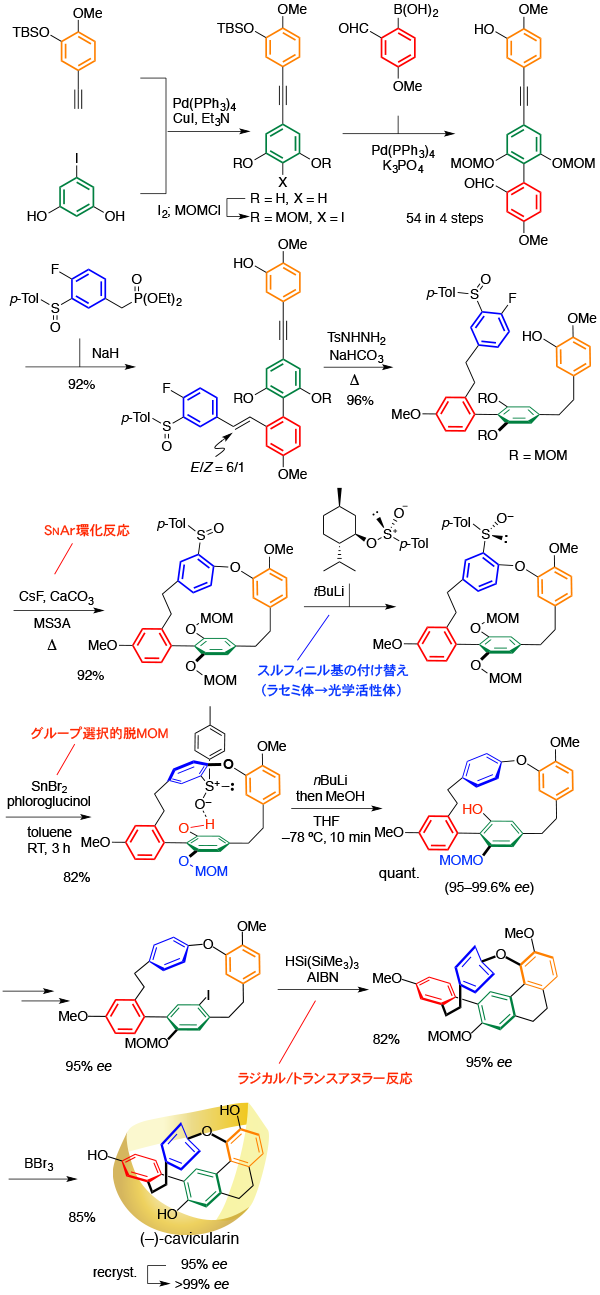
[233] H. Takiguchi, K. Ohmori, K. Suzuki, Angew. Chem. Int. Ed. 2013, 52, 10472.
Total Synthesis of Cinnamtannin B1
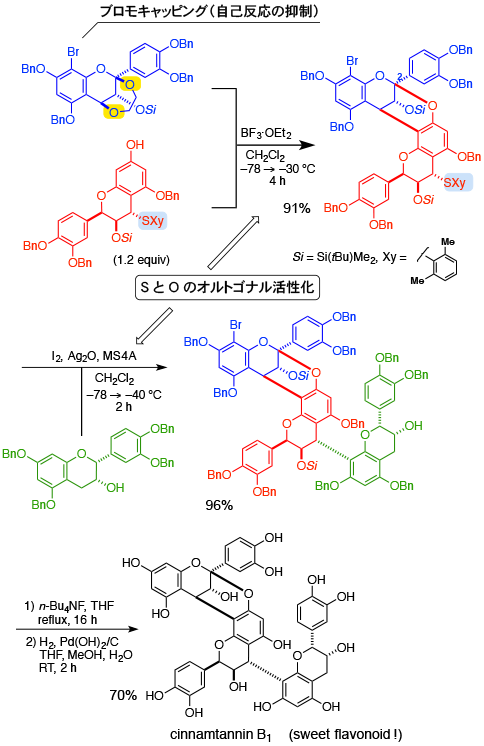
[237] Y. Ito, K. Ohmori, K. Suzuki, Angew. Chem. Int. Ed. 2014, 53, 10129.